Chemistry | Summary and Q&A
TL;DR
This video provides an overview of chemistry, focusing on the periodic table, elements, atoms, molecules, compounds, isotopes, and formulas.
Key Insights
- 🫀 The periodic table organizes elements based on their atomic number and chemical properties.
- 🫀 Atoms and molecules differ in terms of the number of atoms they consist of.
- 📏 Compounds can be molecular or ionic, with different rules for naming and writing their formulas.
- #️⃣ Isotopes have the same number of protons but different numbers of neutrons, leading to different mass numbers.
Transcript
so in this lesson today we're going to focus on chemistry so for those of you who are about to learn chemistry or about to take a chemistry course this video will be very useful for you so the first thing we need to talk about is the periodic table now I recommend that you go to Google images and type in periodic table and print one uh that you cou... Read More
Questions & Answers
Q: What are the names of the elements in Group 1 of the periodic table, and what charge do they form when they become ions?
The elements in Group 1, also known as the alkaline metals, include hydrogen, lithium, sodium, potassium, rubidium, and cesium. When they form ions, they tend to form ions with a positive one charge.
Q: How do you distinguish between an atom and a molecule?
Atoms are individual particles of an element, while molecules consist of two or more atoms bonded together. Elements can exist as atoms or molecules, with molecules containing different types of atoms.
Q: What are the properties of metalloids, and can you provide examples?
Metalloids are elements that have properties in between metals and non-metals. Examples include boron, silicon, and germanium. Metalloids can conduct a small amount of electricity, unlike non-metals.
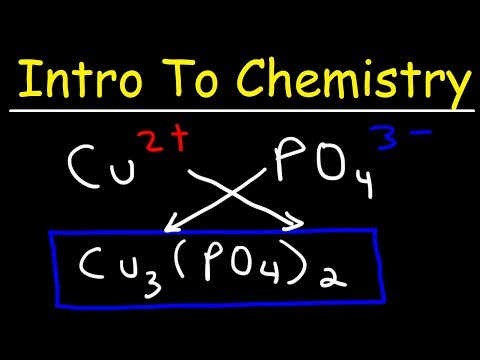
Q: How do you determine the chemical formula of an ionic compound?
To determine the formula of an ionic compound, you need to balance the charges of the ions present. The charges can be determined based on the periodic table and the oxidation states of the elements involved.
Summary & Key Takeaways
-
The video introduces the periodic table and explains the names and characteristics of elements in various groups, such as the alkaline metals, alkaline earth metals, halogens, and noble gases.
-
It distinguishes between atoms and molecules, highlighting the difference between pure elements and compounds.
-
The video discusses the formation and naming of ionic and molecular compounds, including the use of prefixes and Roman numerals.
-
It explains how to determine the number of protons, neutrons, and electrons in an atom or ion, and provides examples of calculations for different elements and isotopes.
Share This Summary 📚
Explore More Summaries from The Organic Chemistry Tutor 📚





